 
The one group in particular that has paved the way is the Thrombolysis in Myocardial Infarction (TIMI) Study Group at Brigham and Women’s Hospital and Harvard University, along with others such as Duke Clinical Research Institute (DCRI) at Duke University, Clinical Trial Service Unit (CTSU) at the University of Oxford, and the Population Health Research Institute at McMaster University. The concept of AROs dates to the 1980s when a few groups started academic international clinical studies. AROs consist of teams of academic investigators aiming to conduct clinical research to address clinical questions and unmet needs, and to educate the next generation of clinical investigators to raise the level of overall clinical research, including clinical trials. The term ARO refers to an academic or nonprofit organization which focuses on developing clinical evidence systematically to improve patient care across the globe. 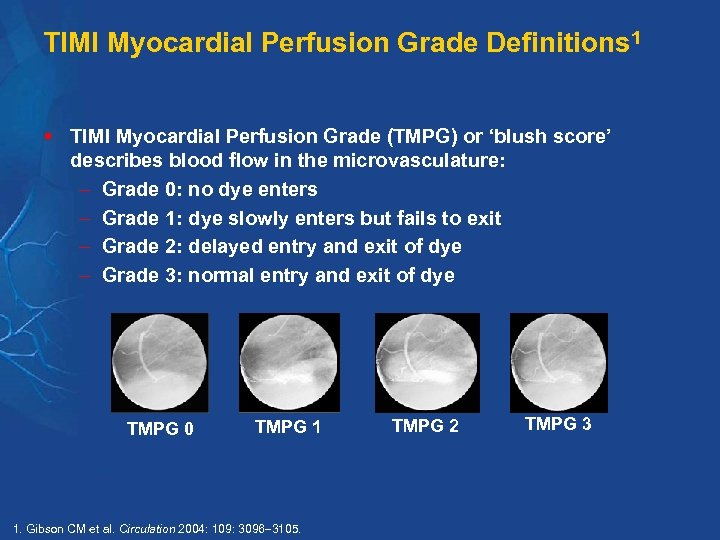 
There has been substantial scientific progress in the understanding of the pathophysiology and treatment of atherosclerotic cardiovascular disease (ASCVD) over the last century, which we owe to decades of basic research and to the upsurge of refined and sophisticated clinical research led by academic research organizations (AROs). In this review article, the authors aim to summarize major research lead by the TIMI Study Group in the ASCVD field. Through a mutual goal to improve the care of ASCVD patients, the Japanese scientific community has become one of the important contributors to the TIMI Study Group’s clinical research. By leading large-scale, international, randomized, controlled trials of novel therapeutics, the TIMI Study Group has helped shape the very practice of cardiovascular medicine for over a quarter of a century, and decades of research continue to provide future promise for further advancement. However, over the years, the TIMI Study Group has expanded their research interests to include antithrombotic therapy, lipid lowering, anti-diabetes, anti-obesity, and even heart failure. The Thrombolysis in Myocardial Infarction (TIMI) Study Group was initially established to conduct a clinical trial studying thrombolysis for treatment of myocardial infarction. This has led to the clinical development of various novel therapeutic interventions for patients with or at risk of ASCVD, in which randomized clinical trials played a crucial role. The elucidation of the atherogenesis mechanism is considered one of the most relevant scientific accomplishments of the last century. The CTO provides contracting, budgeting, and clinical trial management services for Mass General Brigham hospital investigators and industry sponsors and is tasked with supporting and growing clinical trials research at Mass General Brigham.Atherosclerotic cardiovascular disease (ASCVD) is the leading cause of morbidity and mortality across the world, warranting continuous research in this field. Wiviott serves as Vice President for Clinical Trials Research and Administration for Mass General Brigham and leads the Mass General Brigham Clinical Trials Office. He was named in 20 by Thomson Reuters and Clarivate Analytics as one of “The World’s Most Influential Scientific Minds” and in 2016-2020 as a “Highly Cited Researcher,” recognitions inclusive of the top 1% of researchers in Clinical Medicine by citation.īuilding on his trials experience, Dr. Wiviott is an author of over 200 peer-reviewed publications in major medical and cardiovascular journals including 15 original articles in the New England Journal of Medicine. He has also served in leadership of trials of secondary prevention of cardiovascular disease as global PI of DECLARE – TIMI 58, CAMELLIA – TIMI 61, and DAPA ACT HF – TIMI 68 assessing CV safety and efficacy of metabolic therapies. He is the Chairman of the TIMI Clinical Events Committee and recognized as an expert in event definitions and adjudications. Dr. Wiviott has played important roles in the planning, implementation, leadership, and interpretation of multicenter, national, and international clinical trials in acute coronary syndromes including the TRITON-TIMI 38 trial, PRINCIPLE-TIMI 44 and of the TIMI 38 Coronary Stent Registry. 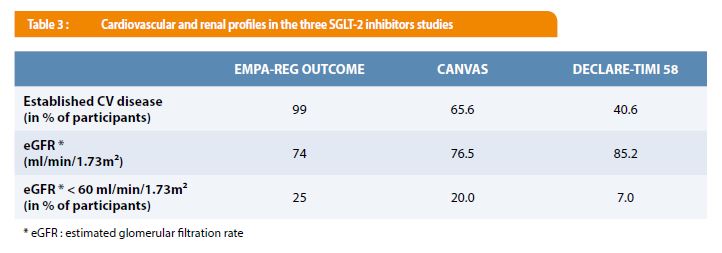
He is a Professor of Medicine at Harvard Medical School, and a Senior Investigator with the TIMI study group.Īs an investigator, Dr. Following his medical residency training, he served as a Cardiology fellow at Johns Hopkins Hospital. He served as a Medicine resident and Chief Medical Resident at Brigham and Women’s Hospital (BWH). Wiviott, MD, FACC is a graduate of University of Pennsylvania and Harvard Medical School (HMS, Honors). Vice President, Clinical Trials Research and Administration 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
